Anticipating the Unexpected: How ChemAIRS Turns Impurity Surprises into a Thing of the Past
Every process chemist knows the feeling. You've optimized your reaction, your yields look great — and then the LC trace comes back with peaks you didn't plan for.
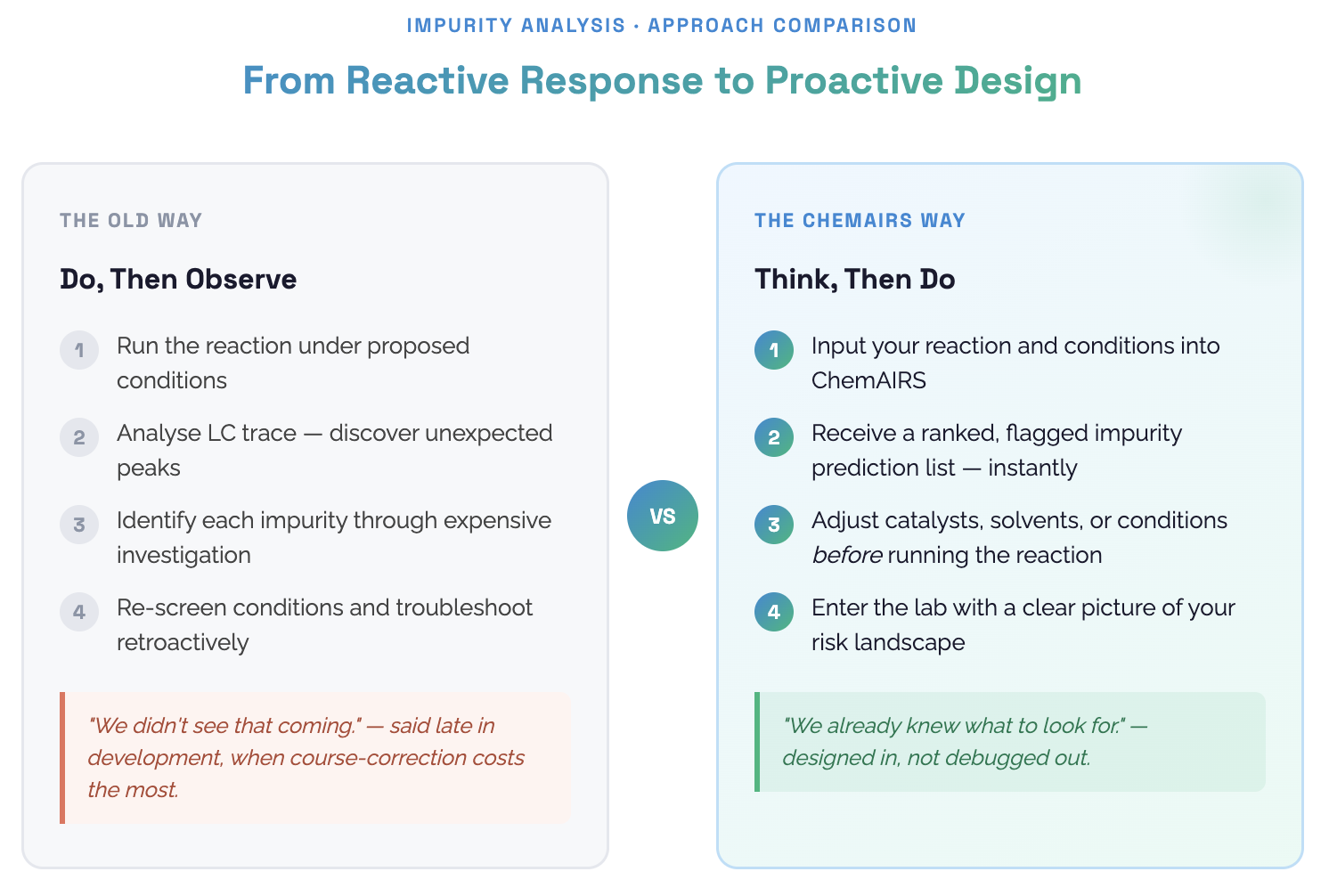
Impurities don't announce themselves. They show up uninvited, late in development, when the cost of course-correcting is highest. The traditional approach? Run the reaction, observe the mess, then work backwards. It's reactive by nature, expensive by consequence.
ChemAIRS was built to change that.
The Real Cost of "We Didn't See That Coming"
Unexpected impurities aren't just a scientific nuisance — they're a business risk. They can trigger regulatory questions, delay IND filings, force late-stage route changes, and in the worst cases, introduce safety liabilities that weren't on anyone's radar.
The challenge is that organic reactions are rarely as clean as the arrow-pushing in a textbook. Every functional group in a molecule is a potential reaction site. Catalysts, solvents, temperature, and moisture — each variable can open a new side pathway. Mapping all of that manually, from first principles, for every reaction step, is exactly the kind of time-consuming expert work that slows development down.
This is the problem ChemAIRS solves — not by replacing chemist intuition, but by supercharging it.
ChemAIRS in Action: Two Real-World Cases
Case Study 1: Fostemsavir — Taming a Complex C–N Coupling
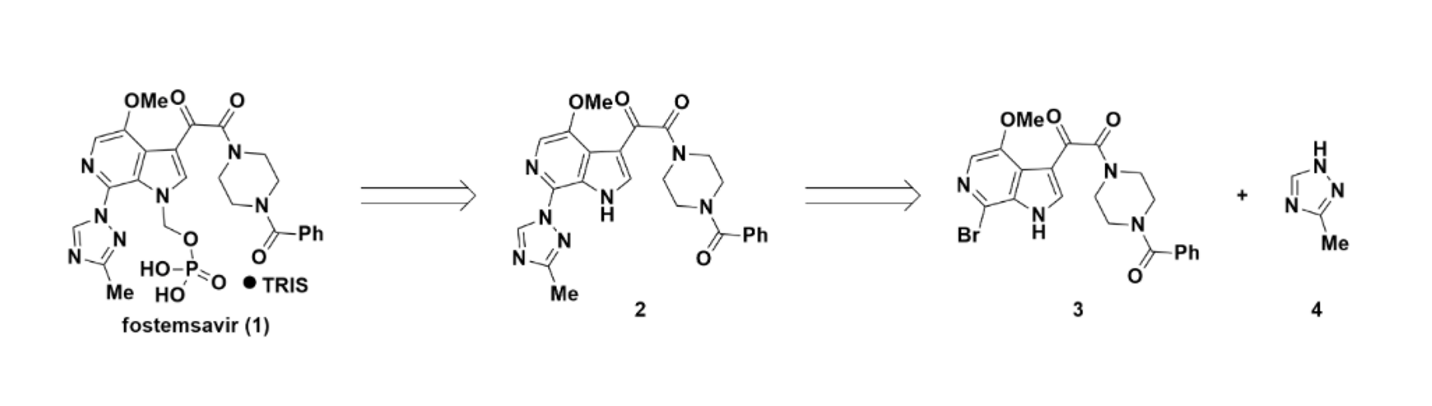
Fostemsavir (1), the HIV-1 attachment inhibitor, presents one of the more demanding coupling challenges in pharmaceutical synthesis: a regioselective C–N coupling between bromoazaindole 3 and 1,2,4-triazole nucleophile 4 (Figure 1).1,2 The triazole can couple through two different nitrogen atoms — and that's before you account for competitive reactions with the ligand, excess base, and the solvent environment.
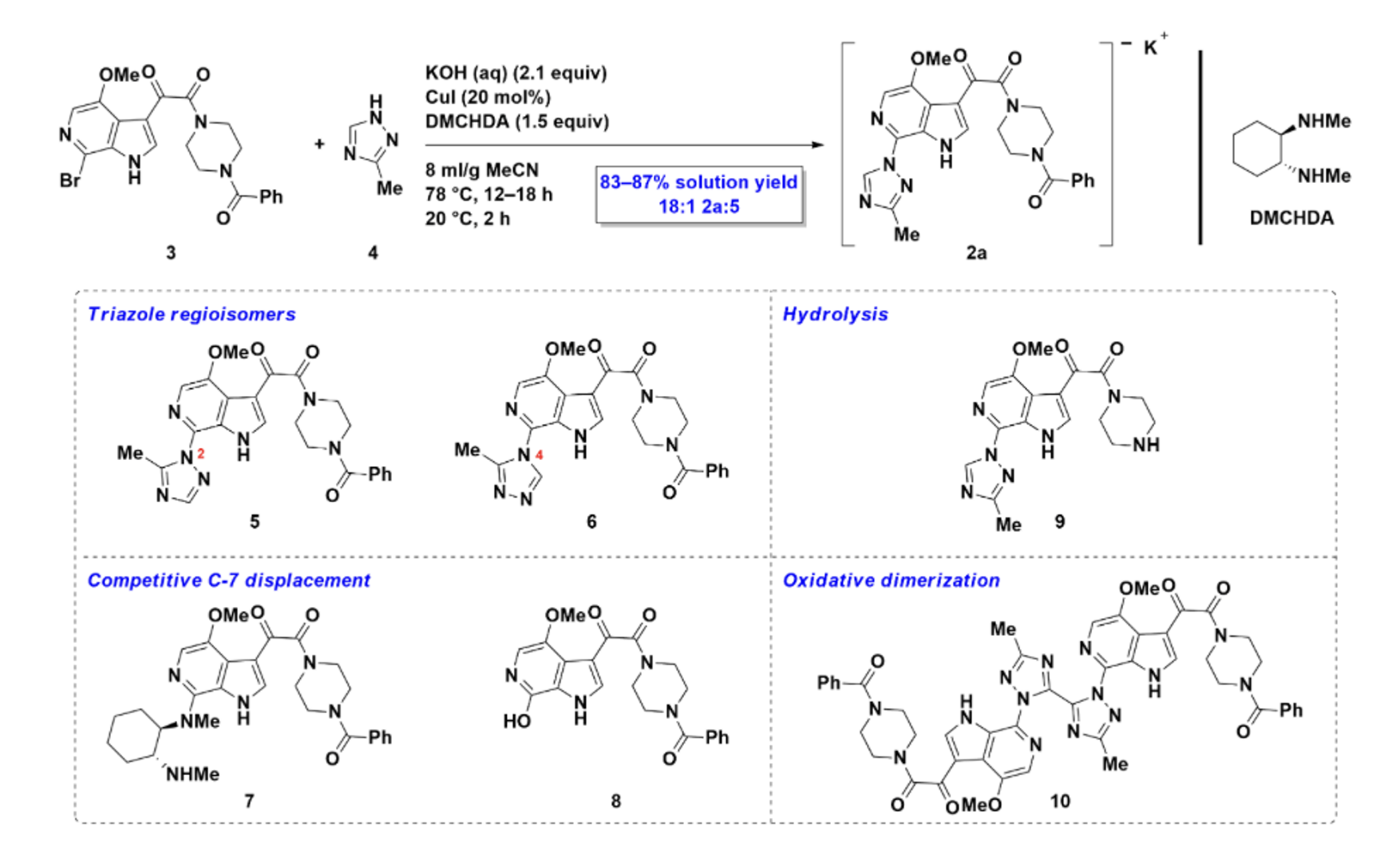
In the published synthesis, six distinct process impurities were identified experimentally — only after extensive condition screening. That's six unknowns that had to be chased down in the lab.
Figure 1. Partial retrosynthetic analysis of fostemsavir (1)
What ChemAIRS did instead:
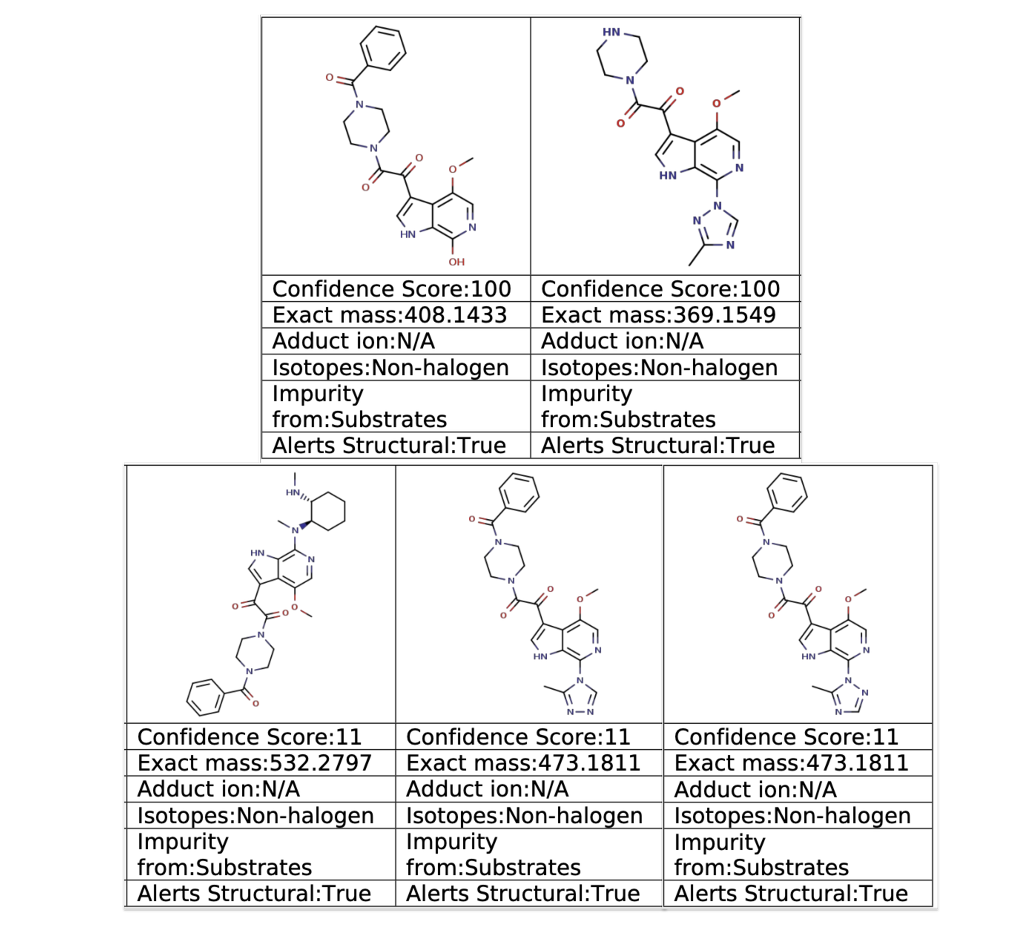
In a single analysis run, ChemAIRS Impurity Prediction identified five of those six impurities — including both triazole regioisomers 5 and 6, the DMCHDA ligand adduct 7, the C-7 hydroxide displacement product 8, and the des-benzoyl hydrolysis product 9 — before a single reaction was run (Figure 2). Each predicted impurity was flagged with a confidence score, exact mass, adduct ion, isotope classification, impurity source, and structural alert flag (Figure 3).
Figure 2. Process impurity profile for the synthesis of potassium salt 2a
Figure 3. Selected results from the ChemAIRS Impurity Prediction analysis of C–N coupling in the synthesis of fostemsavir
The one impurity class ChemAIRS couldn't auto-generate — the oxidative dimerization product 10 — isn't a dead end either. Users can manually add known impurities to the results list, and ChemAIRS will remember that reaction type going forward. The system gets smarter with use.
The competitive edge:
Your team walks into condition screening already knowing which impurities to watch for, which structural alerts to flag for safety review, and which reaction variables are most likely to shift the impurity profile. That's not a minor efficiency gain — that's a fundamentally different way of doing process chemistry.
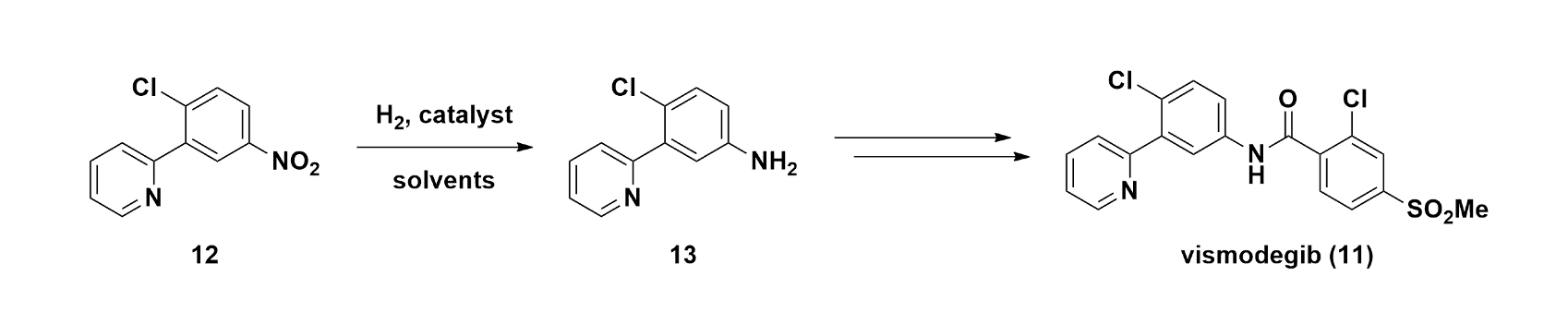
Case Study 2: Vismodegib — Reading the Reaction Before It Runs
The nitro reduction step in the vismodegib (11, Figure 4) synthesis looks deceptively straightforward on paper: reduce a nitroaryl (12) to an aniline (13).3 In practice, the impurity profile is highly sensitive to conditions, and the risks are not trivial.
Figure 4. General scheme for reduction of nitroaryl 12 to aniline 13 and elaboration to vismodegib (11)
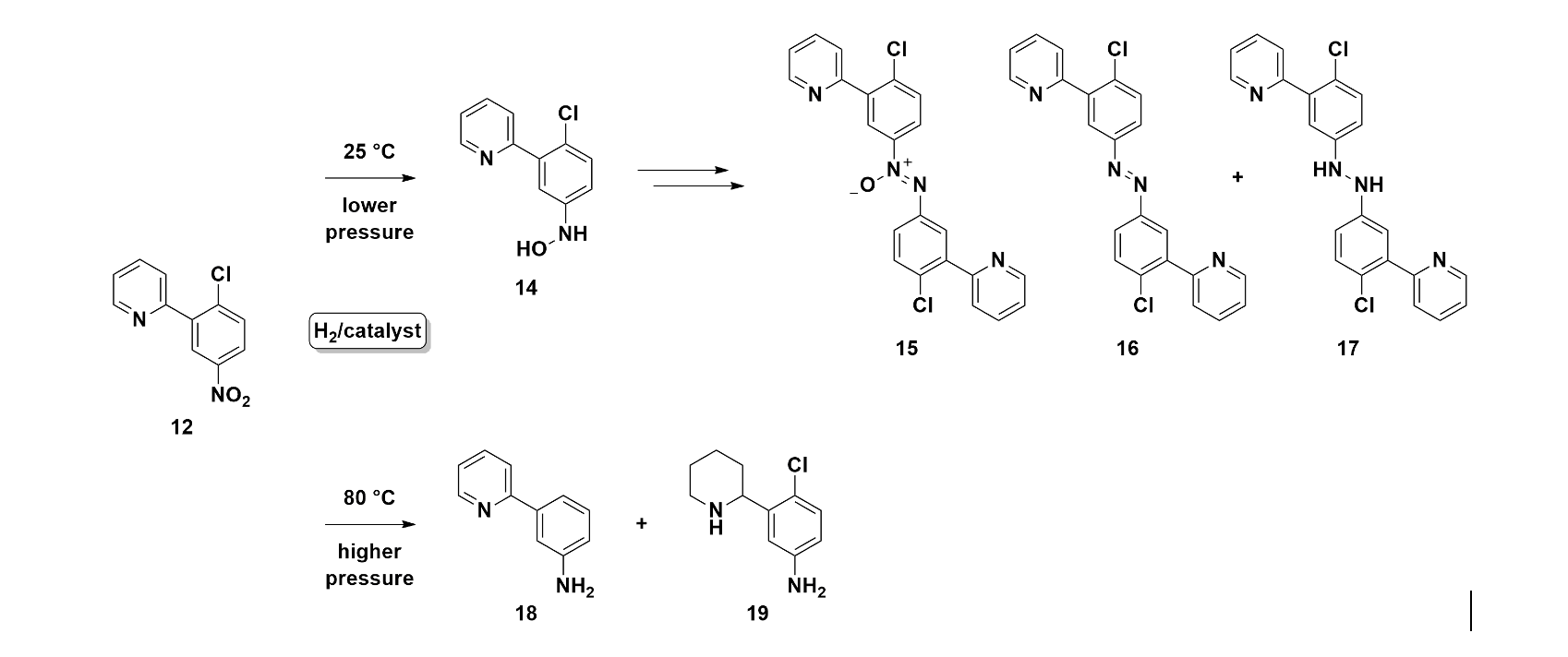
At lower temperatures and pressures, incomplete reduction produces hydroxylamine intermediates (14), azo byproducts (15, 16) from hydroxylamine dimerization, and hydrazine derivatives (17) from further azo reduction (Figure 5). Push the temperature and pressure up, and you get hydrodehalogenation (18) and over-reduction of the ring system (19). Different conditions, entirely different impurity landscapes.
Figure 5. Impurities generated by reduction of nitroaryl 12 under different conditions
What ChemAIRS did:
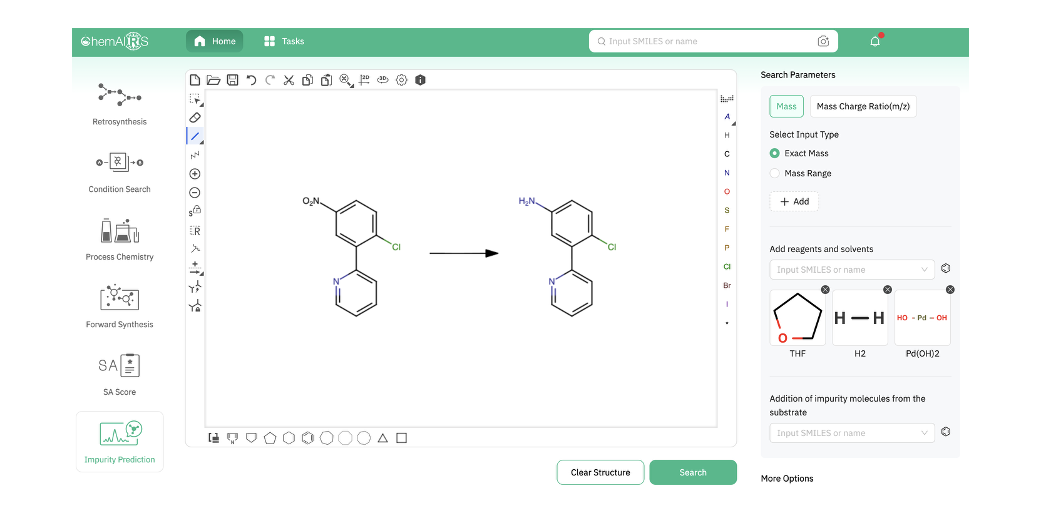
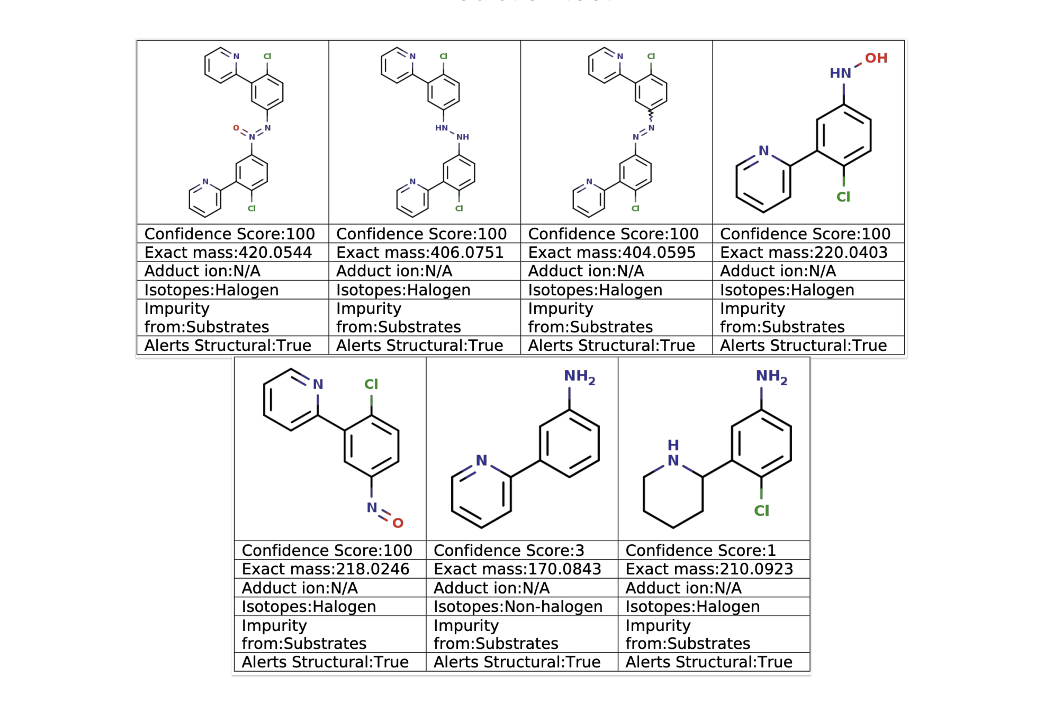
Run through the ChemAIRS Impurity Prediction tool with the reaction conditions specified, and all six experimentally observed impurities 14–19 were captured in the output. But ChemAIRS didn't stop there — it also flagged an additional nitroso compound not present in the published impurity profile, one that carries potential nitrosamine precursor liability.
Figure 6. Input for analysis of the nitroaryl reduction using the ChemAIRS Impurity Prediction tool
Figure 7. Selected results from ChemAIRS Impurity Prediction analysis of nitroaryl reduction
This matters enormously in today's regulatory environment, where nitrosamine risk is under intense scrutiny from the FDA and EMA. ChemAIRS didn't just match what the literature found — it found something the literature missed.
The competitive edge:
Safety-relevant flags, surfaced proactively, before you're in a conversation with a regulator. That's the kind of coverage that protects programs.
From "Do and Observe" to "Think, Then Do"
The pattern across both case studies is the same: ChemAIRS shifts the locus of discovery from the bench to the planning stage.
When you know in advance which side reactions are mechanistically plausible — which functional groups are labile, which pathways are kinetically accessible, how conditions shift the risk profile — you can design around problems rather than troubleshoot your way out of them. Adjust the catalyst. Tighten moisture control. Choose a ligand that doesn't compete. Add an inhibitor. These are decisions that are easy to make early and expensive to make late.
ChemAIRS doesn't replace the chemist's expertise. It gives that expertise a head start.
Why This Is Different?
Most impurity analysis tools in the market are either database lookups — useful only if your impurity has been seen before — or rule-based filters that flag structural alerts without mechanistic context.
ChemAIRS Impurity Prediction reasons from reaction mechanisms. It understands functional group chemistry. It factors in your actual reaction conditions. And it learns from the impurities your team adds to its results, building institutional knowledge that compounds over time.
The result is a tool that works the way a senior process chemist thinks — systematically, mechanistically, and always one step ahead of the reaction.
Ready to see what ChemAIRS finds in your next synthesis step?
References
1. Yan, M.; Baran, P. S. BMS-663068: Another Quiet Victory for Chemistry. Org. Process Res. Dev. 2017, 21 (8), 1091–1094. doi:10.1021/acs.oprd.7b00208
2. Gallagher, W. P.; Soumeillant, M.; Chen, K.; Fox, R. J.; Hsiao, Y.; Mack, B.; Iyer, V.; Fan, J.; Zhu, J.; Beutner, G.; Silverman, S. M.; Fanfair, D. D.; Glace, A. W.; Freitag, A.; Sweeney, J.; Ji, Y.; Blackmond, D. G.; Eastgate, M. D.; Conlon, D. A. Preparation of the HIV Attachment Inhibitor BMS-663068. Part 7. Development of a Regioselective Ullmann–Goldberg–Buchwald Reaction. Org. Process Res. Dev. 2017, 21 (8), 1156–1165. doi:10.1021/acs.oprd.7b00191
3. Angelaud, R.; Reynolds, M.; Venkatramani, C.; Savage, S.; Trafelet, H.; Landmesser, T.; Demel, P.; Levis, M.; Ruha, O.; Rueckert, B., Jaeggi, H. Manufacturing Development and Genotoxic Impurity Control Strategy of the Hedgehog Pathway Inhibitor Vismodegib. Org. Process Res. Dev.2016, 20 (8), 1509–1519. doi:10.1021/acs.oprd.6b00208