Exploring Synthetic Pathways for Deep-Blue OLED Emitters Using ChemAIRS
Deep-Blue OLED Emitters: Overcoming the Last Color Challenge
Deep-blue emitters are a critical component of OLED (organic light-emitting diode) technology. Together with red and green emitters, they complete the RGB primary color set, enabling wide color gamuts and high color purity in modern displays. Among these, deep-blue emission plays a disproportionate role in determining how vibrant and color-accurate a device ultimately appears [1].
Despite decades of progress in OLED materials, blue - and especially deep-blue - emitters remain the most difficult to optimize. Their large bandgaps, required to reach short-wavelength emission, often lead to lower efficiency and significantly shorter operational lifetimes compared with red and green counterparts [2]. High-energy excitons in blue devices accelerate degradation pathways, making blue emitters a primary bottleneck for OLED lifetime and stability.
Conventional Approaches and Their Limitations
A wide range of blue-emitting building blocks has been explored, including benzimidazole (PI), phenanthroimidazole (PPI), and pyrene-imidazole (PyPI) derivatives. These rigid π-conjugated frameworks offer high photoluminescence quantum yields, strong thermal stability, and good film-forming properties - essential requirements for OLED fabrication.
However, despite extensive molecular optimization, deep-blue emitters continue to lag behind. Conventional strategies - primarily based on substituent modification - often fine-tune emission color or efficiency but fail to address a deeper limitation: excited-state dynamics under device operating conditions. As a result, efficiency roll-off and exciton loss processes remain difficult to suppress.
A Different Strategy: Regional Isomerization
A recent study published in Advanced Optical Materials takes a fundamentally different approach [3]. Instead of modifying peripheral substituents, the authors employ regional isomerization, altering the conjugation pathway within the same rigid molecular core.
This subtle structural change has a pronounced effect. By reorganizing electronic communication inside the molecule, regional isomerization modulates excited-state dynamics - reshaping how triplet excitons are dissipated and harvested under device operation.
Molecular Design Context
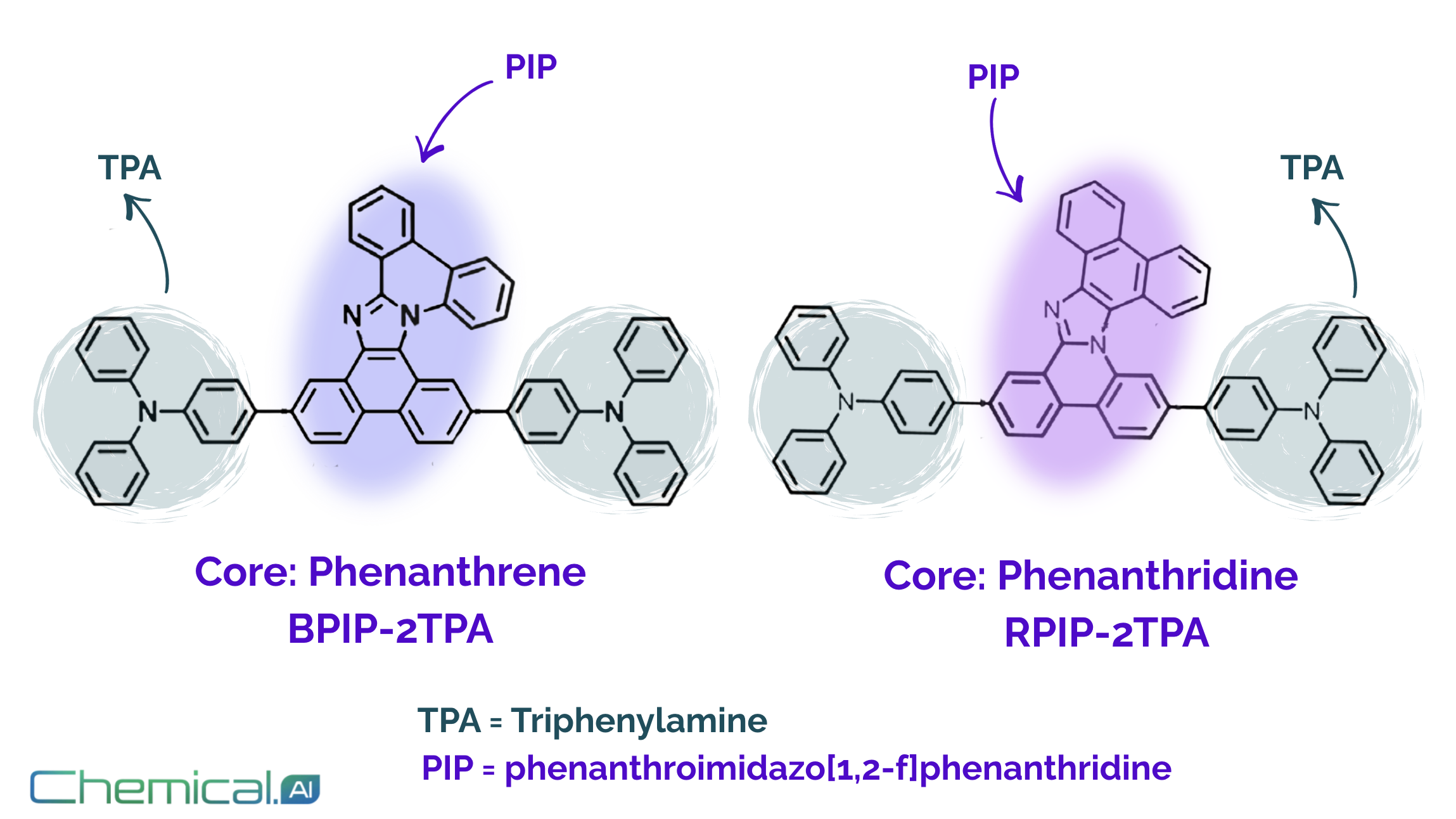
Both BPIP-2TPA and RPIP-2TPA are built on a rigid, helical phenanthroimidazo[1,2-f]phenanthridine (PIP) core, a nitrogen-containing fused polycyclic scaffold known to support hybridized local and charge-transfer (HLCT) excited states. The two emitters share the same molecular formula and triphenylamine (TPA) donor units; their only difference lies in where conjugation is extended along the PIP core (Figure 1).
Figure 1.
In BPIP-2TPA, conjugation proceeds along the phenanthrene side of the core, whereas in RPIP-2TPA it extends along the phenanthridine side, subtly altering the involvement of pyridinic versus pyrrolic nitrogen atoms in the electronic structure. This regional isomerization does not change molecular rigidity or steric profile, but it significantly influences charge-transfer character and high-energy excited-state dynamics - ultimately leading to distinct triplet-exciton utilization pathways and device behavior.
Key Outcomes and Mechanistic Insights
Using the PIP core, the team designed two high-performance deep-blue emitters - BPIP-2TPA and RPIP-2TPA - that exhibit markedly different excited-state behaviors despite their close structural similarity.
Highlights at a Glance
Performance breakthrough
At a practical brightness of 1000 cd m⁻², the optimized BPIP-2TPA-doped OLED maintains an external quantum efficiency (EQE) of 11.02% (EQE<sub>max</sub> = 11.11%), with low efficiency roll-off.
Innovative design strategy
Atomic-level regional isomerization within the helical PIP core tunes the roles of pyridinic versus pyrrolic nitrogen atoms, reshaping charge-transfer contributions and high-energy excited-state dynamics that govern triplet utilization.
Application potential
The results position the PIP core as a powerful platform for developing high-efficiency, low-roll-off deep-blue OLED emitters through deliberate excited-state pathway engineering.
Collectively, the study shows that the PIP core is not only an efficient emissive unit but also a highly tunable structural scaffold, offering a new molecular design dimension for next-generation deep-blue OLED materials.
Enabling Synthesis Through Flexible Route Design
Translating advanced emitter designs into real molecules requires practical and adaptable synthetic routes. For complex molecules such as RPIP-2TPA, careful route planning is essential to balance feasibility, cost, and scalability.
Using ChemAIRS retrosynthesis, three distinct synthetic routes were identified for RPIP-2TPA.
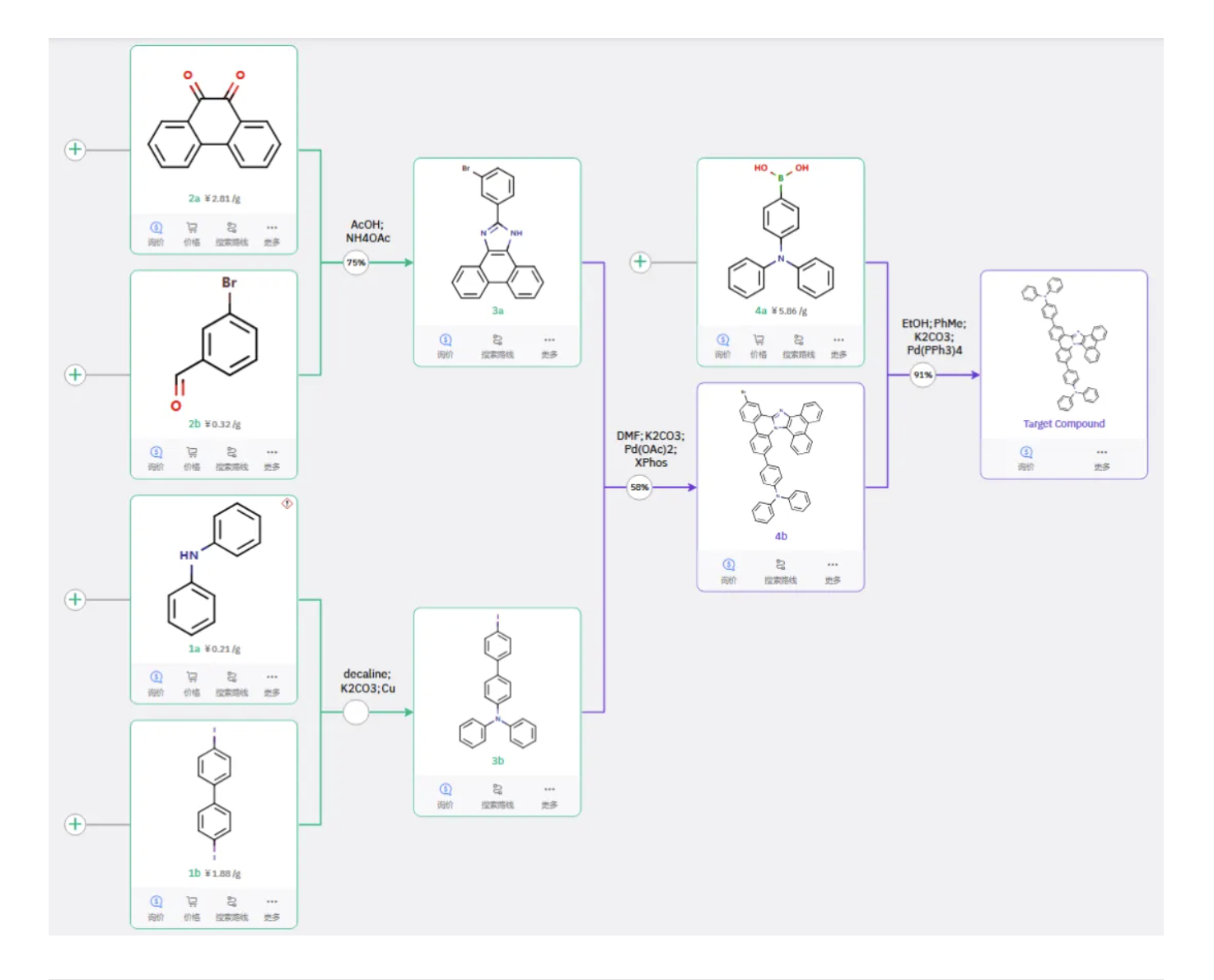
Route 1
Starting materials 1a and 1b undergo Ullmann coupling to give intermediate 3b. Starting materials 2a and 2b undergo ring closure to form 3a. Intermediates 3a and 3b undergo Buchwald C–N coupling to afford 4b, while 4a reacts with 4a via Suzuki coupling to yield the target product.
Route 1.
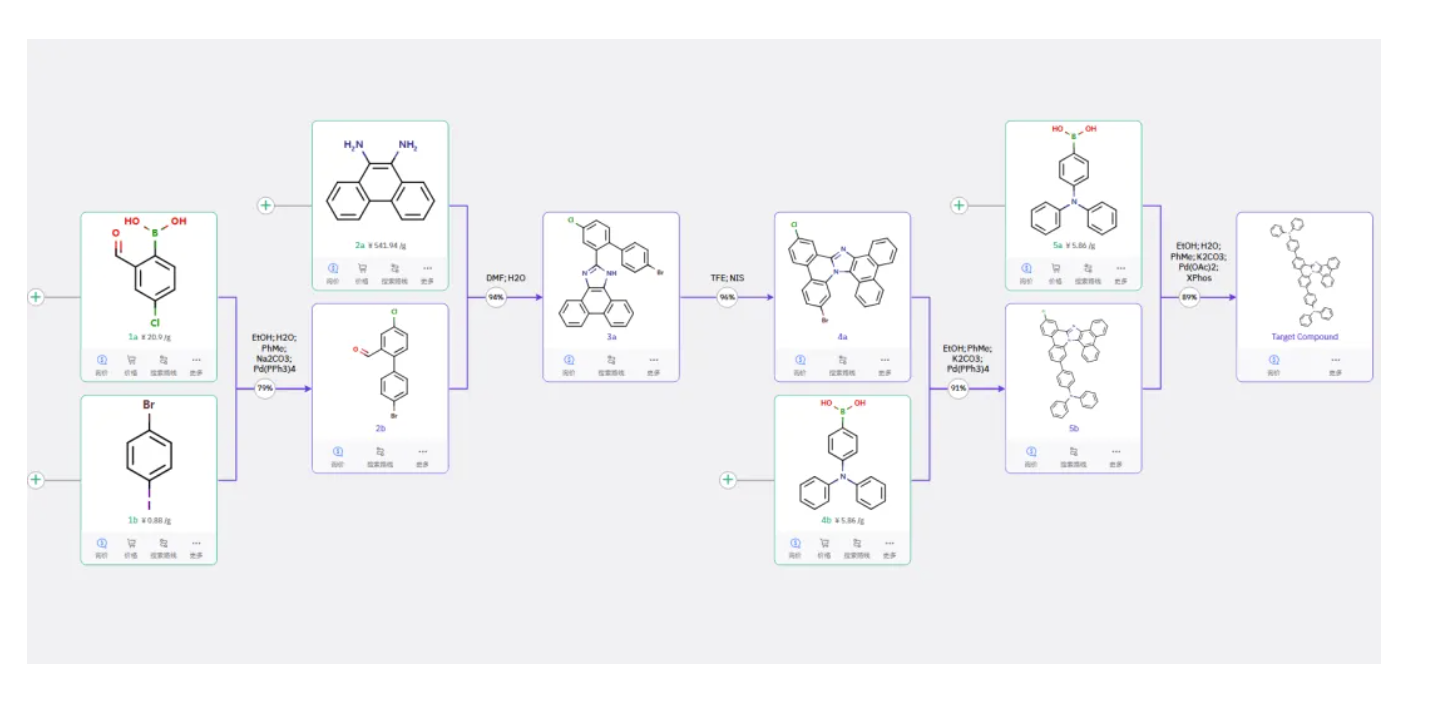
Route 2
Starting materials 1a and 1b undergo Suzuki coupling to form 2b, which reacts with 2a to close the imidazole ring and form 3a. Further intramolecular cyclization yields 4a, which then undergoes a one-pot Suzuki coupling with boronic acids 4b and 5a to afford the target product.
Route 2.
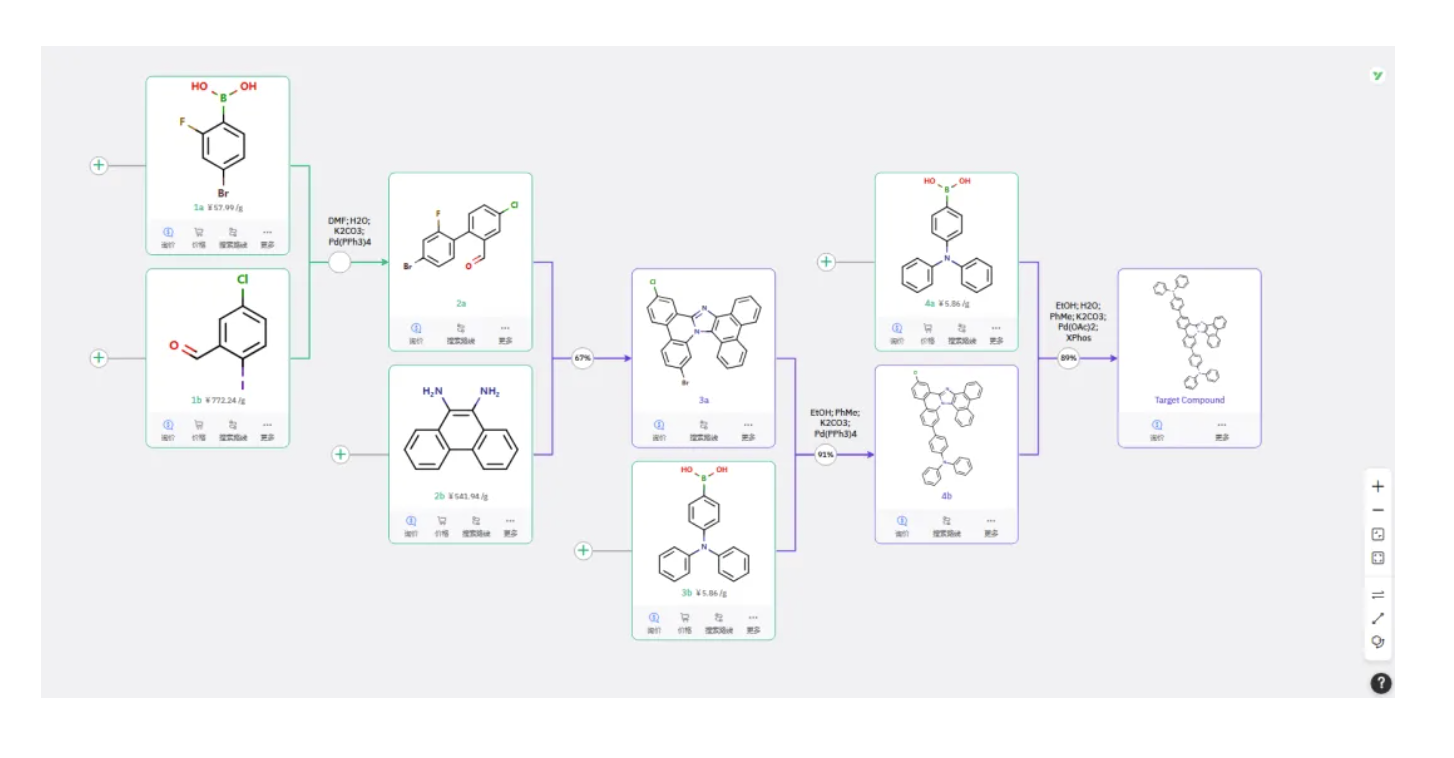
Route 3
Boronic acid 1a and iodide 1b undergo Suzuki coupling to form 2a, which then reacts with 2b to undergo two consecutive intramolecular ring-closure reactions, yielding 3a. Subsequent Suzuki coupling with a boronic acid produces 4b, followed by coupling with 4a to afford the target product.
Route 3.
Rather than prescribing a single optimal pathway, ChemAIRS enables rapid comparison of viable strategies, allowing chemists to adapt synthesis plans as materials development progresses. This flexibility accelerates experimentation and supports more robust OLED materials R&D workflows.
References
[1] S. Sreejith et al., Micro and Nanostructures, 200 (2025).
[2] I. D. W. Samuel, E. Zysman-Colman, J. Phys. Chem. Lett., 15 (2024), 1034–1047.
[3] Y. Huang, C. Xiao, J. Lou, et al., Advanced Optical Materials (2025), e02266.